MCAT Practice Questions: Physics
Walking into MCAT preparation, many students think of physics as the least applicable science to medicine, reflecting on calculus-heavy premedical classes. But even in the medical field, physics is all around us. When we treat patients at a rehab hospital, we often talk about motion, forces, and bone strength. An ophthalmologist may draw diagrams to help students better understand myopia and hyperopia. When we talk about mitochondria functioning as the batteries of the cell, we mean that fairly literally.
MCAT Practice Questions: Physics
MCAT Physics Practice Questions: Explained
MCAT Physics Question #1 Explained
Using the Pythagorean theorem, calculate the magnitude of the man’s displacement:
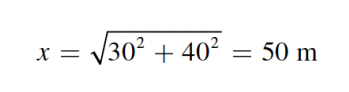
His total distance traveled is equal to 30 + 40 = 70 m. Therefore, the difference between these two is 20 m.
MCAT Physics Question #2 Explained
In order for the seesaw to be balanced, the torque due to the girl (τg ) must be exactly counteracted by the torque due to her father (τf ). In other words, the magnitudes of these torques must be equal (τg = τf ):
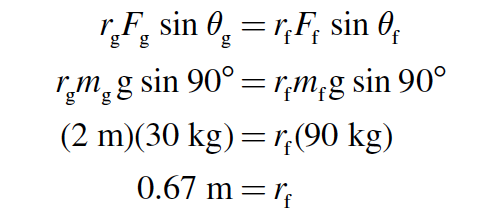
Because r represents the distance of each person from the fulcrum, the father must sit 67 cm from the fulcrum.
Although the designers of the MCAT will provide physiological facts and numbers, that’s not what you’re expected to know before taking the exam. Your MCAT practice will require being able to see the same concepts that you learned in your undergraduate pre-med classes in a very defined and isolated environment—applied in a foreign scope to integrated sciences.
The interplay of scientific disciplines (i.e., how your knowledge of physics or chemistry informs your understanding of how an organ works) is paramount to your success both on the new MCAT and as a future physician. The human body is a network of interdependent physical, chemical, and biological processes, and the MCAT measures your ability to make those connections.
Knowing formulas and reactions is necessary for success on the MCAT, but will in no way be sufficient. Success will only come with practice, so here are a couple of tips:
MCAT Physics Tip 1: When studying a “physical” science, think through all of its biological applications.
For example, let’s use reduction and oxidation. We know that LEO the Lion says GER (the Loss of an Electron is Oxidation and the Gain of an Election is Reduction). Therefore, when a metal is losing or gaining electrons, it’s either getting oxidized or reduced, respectively.
Don’t stop there. The ReDox that occurs in the Electron transport chain, with NADH losing an electron to the ETC and getting—you guessed it—oxidized, is the exact same concept. NAD+ is the product of the oxidation of NADH, just as Ag+ is the oxidized product of Ag. It’s the exact same science. So, don’t get thrown off by the fact that you learned it in two different places.
MCAT Physics Tip 2: There are a finite number of scientific facts.
There is some truth in the claim that most of biology is rooted in chemistry and physics. Take proteins for example. We think of them as biological molecules because they serve such a prominent role in the body. In reality, they are nothing more than a very specific structural arrangement of carbon, nitrogen, oxygen, hydrogen, and sometimes sulfur.
The way in which they are bonded to each other is through a bonding orbital—just like the ones you learned about in general chemistry. They fold into specific shapes because of attractions and repulsions of the amino acids in their sequence. Those are the same attractions and repulsions seen in chemistry. The proteins themselves are coded from RNA, which is coded from DNA. RNA and DNA are just chemical molecules with the same properties you learned about in chemistry.

